| View previous topic :: View next topic |
| Author |
Message |
dontgogreen
Joined: 02 Aug 2015
Posts: 79



|
 Posted: Dec 15, 2017 08:59 Post subject: Re: Collection of Dontgogreen Posted: Dec 15, 2017 08:59 Post subject: Re: Collection of Dontgogreen |
|
|
After spending a little time collecting worldwide minerals without any focus, I have narrowed my interest to rutile crystals. I think their beauty is a bit more subtle than many species, and they are generally far more affordable. Here are two Brazilian specimens that I like quite a bit.
| Mineral: | Rutile (variety struverite) |
| Locality: | | Golconda pegmatite, Governador Valadares, Vale do Rio Doce, Minas Gerais, Brazil |  |
|
| Dimensions: | ~4.5 cm |
| Description: |
|
| Viewed: |
18009 Time(s) |

|
| Mineral: | Rutile |
| Locality: | | Capão do Lana Mine, Rodrigo Silva District, Ouro Preto, Minas Gerais, Brazil |  |
|
| Dimensions: | 5.5 cm |
| Description: |
|
| Viewed: |
18020 Time(s) |

|
|
|
| Back to top |
|
 |
dontgogreen
Joined: 02 Aug 2015
Posts: 79



|
 Posted: Dec 22, 2017 09:26 Post subject: Re: Collection of Dontgogreen Posted: Dec 22, 2017 09:26 Post subject: Re: Collection of Dontgogreen |
|
|
Hematite and rutile both posses a hexagonal close-packed oxygen sublattice, which enables simple transitions among them by exsolution, epitaxy, and intergrowth (Daneu et al. [2014] and references therein).
Other examples of hematite and rutile intergrowth are the speicmens from Novo Horizonte, Brazil, and those of Mwinilunga, Zambia. A study of the Zambian material indicates that the source of Ti for rutile growth is the hematite crystal itself. Diffusion of Ti ions lead to topotaxial nucleation and growth of rutile on structurally compatible lattice planes (Rečnik et al., 2015). I am not sure whether or not this also holds true for specimens from Cavradi, as I have not been able to find a similar study. If anyone is aware of one, please direct me to it. Additionally, if you have Mwinilunga specimens, I would gladly trade for them!
| Mineral: | Hematite with Rutile |
| Locality: | | Cavradi, Curnera Valley, Tujetsch (Tavetsch), Vorderrhein Valley, Grischun (Grisons; Graubünden), Switzerland |  |
|
| Dimensions: | ~4 cm |
| Description: |
|
| Viewed: |
17914 Time(s) |

|
|
|
| Back to top |
|
 |
Philippe Durand
Joined: 10 May 2016
Posts: 628
Location: Normandie



|
 Posted: Dec 22, 2017 10:23 Post subject: Re: Collection of Dontgogreen Posted: Dec 22, 2017 10:23 Post subject: Re: Collection of Dontgogreen |
|
|
Ha ! I love this hematite; my favorite mineral.
It is a marvelous specimen.
|
|
| Back to top |
|
 |
Tobi
Site Admin

Joined: 07 Apr 2009
Posts: 4108
Location: Germany



|
 Posted: Dec 22, 2017 13:35 Post subject: Re: Collection of Dontgogreen Posted: Dec 22, 2017 13:35 Post subject: Re: Collection of Dontgogreen |
|
|
| dontgogreen wrote: | Hematite with rutile
Cavradi, Curnera Valley, Tujetsch (Tavetsch), Vorderrhein Valley, Grischun (Grisons; Graubünden), Switzerland
~4 cm! |
Great Cavradi specimen!!!
|
|
| Back to top |
|
 |
Jordi Fabre
Overall coordinator of the Forum

Joined: 07 Aug 2006
Posts: 4897
Location: Barcelona



|
 Posted: Dec 23, 2017 16:52 Post subject: Re: Collection of Dontgogreen Posted: Dec 23, 2017 16:52 Post subject: Re: Collection of Dontgogreen |
|
|
| dontgogreen wrote: | | ...A study of the Zambian material indicates that the source of Ti for rutile growth is the hematite crystal itself. Diffusion of Ti ions lead to topotaxial nucleation and growth of rutile on structurally compatible lattice planes (Rečnik et al., 2015)... |
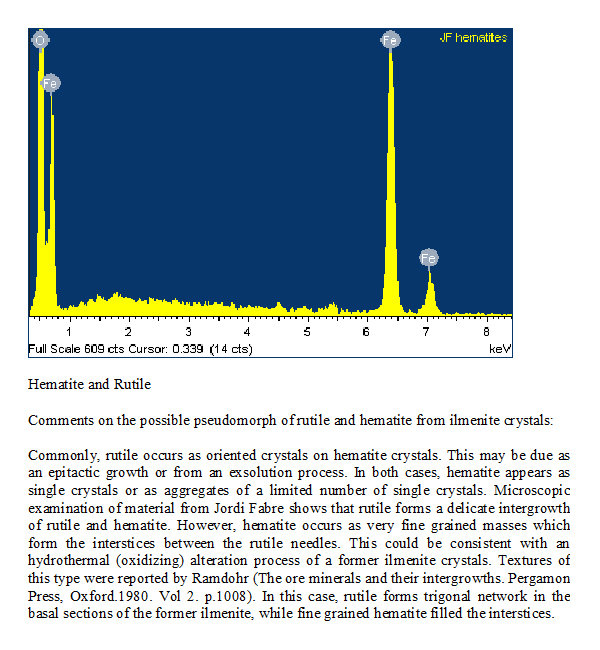
Before the text mentioned it was this note related with the first analysis of the Zambian material wrote in December 2007 by the late Dr. Joan Viñals :
Comments on the possible pseudomorph of rutile and hematite from ilmenite crystals:
Commonly, rutile occurs as oriented crystals on hematite crystals. This may be due as an epitactic growth or from an exsolution process. In both cases, hematite appears as single crystals or as aggregates of a limited number of single crystals. Microscopic examination of material from Jordi Fabre shows that rutile forms a delicate intergrowth of rutile and hematite. However, hematite occurs as very fine grained masses which form the interstices between the rutile needles. This could be consistent with an hydrothermal (oxidizing) alteration process of a former ilmenite crystals. Textures of this type were reported by Ramdohr (The ore minerals and their intergrowths. Pergamon Press, Oxford.1980. Vol 2. p.1008). In this case, rutile forms trigonal network in the basal sections of the former ilmenite, while fine grained hematite filled the interstices.
| Description: |
| The original analysis and report written by the missed Dr. Joan Viñals |
|
| Viewed: |
17763 Time(s) |
|
|
|
| Back to top |
|
 |
dontgogreen
Joined: 02 Aug 2015
Posts: 79



|
 Posted: Jan 12, 2018 23:38 Post subject: Re: Collection of Dontgogreen Posted: Jan 12, 2018 23:38 Post subject: Re: Collection of Dontgogreen |
|
|
The hoppered termination lets the red color come through nicely at the top. These striated prisms are some of my favorites.
| Mineral: | Rutile |
| Locality: | | Hiddenite, Alexander County, North Carolina, USA |  |
|
| Dimensions: | 2.7 cm |
| Description: |
|
| Viewed: |
17583 Time(s) |

|
| Mineral: | Rutile |
| Locality: | | Hiddenite, Alexander County, North Carolina, USA |  |
|
| Dimensions: | 2.7 cm |
| Description: |
|
| Viewed: |
17568 Time(s) |

|
|
|
| Back to top |
|
 |
dontgogreen
Joined: 02 Aug 2015
Posts: 79



|
 Posted: Jan 13, 2018 23:28 Post subject: Re: Collection of Dontgogreen Posted: Jan 13, 2018 23:28 Post subject: Re: Collection of Dontgogreen |
|
|
A multiply twinned equant crystal from Graves Mountain with excellent luster.
| Mineral: | Rutile |
| Locality: | | Graves Mountain, Lincoln County, Georgia, USA |  |
|
| Dimensions: | 2.5 cm |
| Description: |
|
| Viewed: |
17523 Time(s) |

|
|
|
| Back to top |
|
 |
dontgogreen
Joined: 02 Aug 2015
Posts: 79



|
 Posted: May 03, 2018 15:44 Post subject: Re: Collection of Dontgogreen Posted: May 03, 2018 15:44 Post subject: Re: Collection of Dontgogreen |
|
|
Here is an interesting multiply-twinned crystal reported to be from Zagi Mountain. I will produce photos of the full specimen at a later date, but for now I am interested to explore the twin boundaries. Daneu et al. (2014) studied the structure and formation of (101) rutile twins from Diamantina and in this paper, which I do not purport to fully comprehend, the authors describe a complex process by which these twins form. They propose that the formation of these twins is chemically induced, and a result of topotaxial replacement of an Al-rich oxyhydroxide precursor. They find corundumn precipitates segregated on the (101) twin boundary, suggesting that they are remnants of past transient processes involving the dehydration of a parent Al-rich hydroxlian pseudorutile phase, which served as a precursor for rutile crystallization at elevated temperatures. During the late stages of dehydration, the transformation of aforementioned corundumn particles from diaspore leads to a volume decrease, causing tensions in the surrounding rutile which are compensated by the formation of parallel {101} twin boundaries. This is really quite a fascinating paper and I suggest that you read it, as I am sure that I have missed quite a bit of the story here.
Obviously this crystal is not from Diamantina, but I wonder if the same chemical and temperature-related mechanisms operate to produce the same type of twin? One feature that I note in the photograph is that some of the smaller twins in the center of the photo do not extend along the length of the crystal face, but rather taper off about halfway. This seems to be what one might expect if they are parallel twin boundaries created by a dehydration-induced tension in the crystal structure.
Surely there must be some crystallographers who have something to add?
The citation for the paper I referenced is:
Daneu, N., Recnik, A., and Mader, W., 2014. Atomic structure and formation mechanism of (101) rutile twins from Diamantina (Brazil). American Mineralogist, Volume 99, pages 612-624.
| Mineral: | Rutile |
| Locality: | | Zagi Mountain, Hameed Abad Kafoor Dheri, Peshawar, Khyber Pakhtunkhwa, Pakistan |  |
|
| Dimensions: | FOV ~ 1.5cm |
| Description: |
|
| Viewed: |
16911 Time(s) |

|
|
|
| Back to top |
|
 |
dontgogreen
Joined: 02 Aug 2015
Posts: 79



|
 Posted: May 04, 2018 09:18 Post subject: Re: Collection of Dontgogreen Posted: May 04, 2018 09:18 Post subject: Re: Collection of Dontgogreen |
|
|
Here is the entire specimen. Unfortunately not entirely free from damage, but there are rather few references to large, good quality rutiles from this locality in the literature.
| Mineral: | Rutile |
| Locality: | | Zagi Mountain, Hameed Abad Kafoor Dheri, Peshawar, Khyber Pakhtunkhwa, Pakistan |  |
|
| Dimensions: | 4.3 cm |
| Description: |
|
| Viewed: |
16855 Time(s) |

|
|
|
| Back to top |
|
 |
dontgogreen
Joined: 02 Aug 2015
Posts: 79



|
 Posted: May 23, 2018 10:24 Post subject: Re: Collection of Dontgogreen Posted: May 23, 2018 10:24 Post subject: Re: Collection of Dontgogreen |
|
|
Here is another thumbnail from Graves Mountain; it is similar in character to the thumbnail posted previously but lacking matrix. It is multiply twinned and the re-entrant angle may be seen at the top of the crystal. The forms are almost identical to those of the crystal posted above, but exhibit slightly different degree of development. The price from the 1970's label is $27.50!
| Mineral: | Rutile |
| Locality: | | Graves Mountain, Lincoln County, Georgia, USA |  |
|
| Dimensions: | 2.5 cm |
| Description: |
|
| Viewed: |
16566 Time(s) |

|
|
|
| Back to top |
|
 |
dontgogreen
Joined: 02 Aug 2015
Posts: 79



|
 Posted: Nov 01, 2018 10:50 Post subject: Re: Collection of Dontgogreen Posted: Nov 01, 2018 10:50 Post subject: Re: Collection of Dontgogreen |
|
|
Oh dear, this is not a rutile... but I feel that an ongoing Tungsten project at work is a good enough excuse to stock up on ferberite! It's still black, but definitely not ugly. And to balance it out, a large and multiply-twinned rutile from Graves Mountain. Looking at the complex form, my wife commented that it looks like a fight between a triangle and a square. The winner, in this case, is the collector.
| Mineral: | Ferberite |
| Locality: | | Yaogangxian Mine, Yizhang, Chenzhou Prefecture, Hunan Province, China |  |
|
| Dimensions: | 4.5 cm |
| Description: |
|
| Viewed: |
15168 Time(s) |

|
| Mineral: | Rutile |
| Locality: | | Graves Mountain, Lincoln County, Georgia, USA |  |
|
| Dimensions: | 6.5 cm |
| Description: |
|
| Viewed: |
15187 Time(s) |

|
|
|
| Back to top |
|
 |
Tobi
Site Admin

Joined: 07 Apr 2009
Posts: 4108
Location: Germany



|
 Posted: Nov 02, 2018 03:13 Post subject: Re: Collection of Dontgogreen Posted: Nov 02, 2018 03:13 Post subject: Re: Collection of Dontgogreen |
|
|
| Beautiful specimens and great photo quality, I hope this series will be continued :-)
|
|
| Back to top |
|
 |
dontgogreen
Joined: 02 Aug 2015
Posts: 79



|
 Posted: Nov 02, 2018 07:51 Post subject: Re: Collection of Dontgogreen Posted: Nov 02, 2018 07:51 Post subject: Re: Collection of Dontgogreen |
|
|
| Thank you. I will certainly continue, although the limiting factor here is actually acquiring new minerals. So, stick around for a few decades and watch the collection grow.
|
|
| Back to top |
|
 |
|



