| View previous topic :: View next topic |
| Author |
Message |
Mark Ost

Joined: 18 Mar 2013
Posts: 516
Location: Virginia Beach



|
 Posted: Mar 02, 2014 17:41 Post subject: Re: Daylight Fluorescent Hyalite Opal from Mexico Posted: Mar 02, 2014 17:41 Post subject: Re: Daylight Fluorescent Hyalite Opal from Mexico |
|
|
| Ok, so maybe it's not our fingers but you never know about low level radiation! Jordi may have to support my five eyed children.
|
|
| Back to top |
|
 |
Mark Ost

Joined: 18 Mar 2013
Posts: 516
Location: Virginia Beach



|
 Posted: Mar 02, 2014 17:53 Post subject: Re: Daylight Fluorescent Hyalite Opal from Mexico Posted: Mar 02, 2014 17:53 Post subject: Re: Daylight Fluorescent Hyalite Opal from Mexico |
|
|
| Now that you mention it Pierre, it occurred to me I had a beryl from the same location with opal and it seems to do the same thing yours does. Much more subtle than the Mexican opal but it too reacts in daylight.
|
|
| Back to top |
|
 |
Mark Ost

Joined: 18 Mar 2013
Posts: 516
Location: Virginia Beach



|
 Posted: Mar 02, 2014 18:03 Post subject: Re: Daylight Fluorescent Hyalite Opal from Mexico Posted: Mar 02, 2014 18:03 Post subject: Re: Daylight Fluorescent Hyalite Opal from Mexico |
|
|
Pete
I just checked my Namibian beryl (aquamarine) / schorl/ opal. Under incandescent light it appears a frosted white with no hint of green color. In the late evening light it takes on a green hue. Glass does not seem to filter the effect. I will try in direct sunlight tomorrow. Fluorescent light seems to wash it out. Standing away from a south facing window the green is evident but washes out closer to the window due to brightness. Very responsive to SW, MW even in daylight. Less so LW.
|
|
| Back to top |
|
 |
alfredo
Site Admin

Joined: 30 Jan 2008
Posts: 979



|
 Posted: Mar 02, 2014 18:38 Post subject: Re: Daylight Fluorescent Hyalite Opal from Mexico Posted: Mar 02, 2014 18:38 Post subject: Re: Daylight Fluorescent Hyalite Opal from Mexico |
|
|
| I tried to measure the radioactivity of daylight fluorescent opal with a geiger counter and got nothing above background. Ditto for highly fluorescent artificial "uranium glass". The amount of uranium present is simply too low. As for the dangers of low-level radiation, remember that we are surrounded by it all the time; a specimen of daylight fluorescent uranyl-bearing hyalite seems to add no appreciable quantity to that level. Nothing to worry about.
|
|
| Back to top |
|
 |
Cesar M. Salvan
Site Admin
Joined: 09 Jun 2008
Posts: 126
Location: Alcalá de Henares



|
 Posted: Mar 02, 2014 19:01 Post subject: Re: Daylight Fluorescent Hyalite Opal from Mexico Posted: Mar 02, 2014 19:01 Post subject: Re: Daylight Fluorescent Hyalite Opal from Mexico |
|
|
| As I said in my report, the uranium content can be detected only by sensitive instruments. Hand-held Geiger monitors are not adequate to detect it. GM monitors equipped with thin window detectors, used correctly, can detect the small amount of activity of these samples, as are too close to background.
|
|
| Back to top |
|
 |
Jesse Fisher

Joined: 18 Mar 2009
Posts: 629
Location: San Francisco



|
 Posted: Mar 02, 2014 23:17 Post subject: Re: Daylight Fluorescent Hyalite Opal from Mexico Posted: Mar 02, 2014 23:17 Post subject: Re: Daylight Fluorescent Hyalite Opal from Mexico |
|
|
| At the TGMS show I measured the activity level of a specimen using a hand-held scintallometer that was available in the GSA booth in the convention center lobby. The measured activity level was 1-3 microSieverts per hour and 0. - 0.3 milliRoentgens per hour. According to references, the former measurement is about the some as the exposure one will receive while flying in a typical airliner at cruising altitude (around 40,000 feet). I'm not sure how many hours I have logged flying at 40,000 feet in the many trips between California and London over the past 20 years, but I am sure it is far more than I would ever spend holding one of these specimens, no matter how pretty they are!
|
|
| Back to top |
|
 |
Cesar M. Salvan
Site Admin
Joined: 09 Jun 2008
Posts: 126
Location: Alcalá de Henares



|
 Posted: Mar 09, 2014 16:11 Post subject: Re: Daylight Fluorescent Hyalite Opal from Mexico Posted: Mar 09, 2014 16:11 Post subject: Re: Daylight Fluorescent Hyalite Opal from Mexico |
|
|
I received a lot of questions by email and even by phone, about this opal and its uranium content. Two of these questions are 'faq', so I decided to share it here for general information:
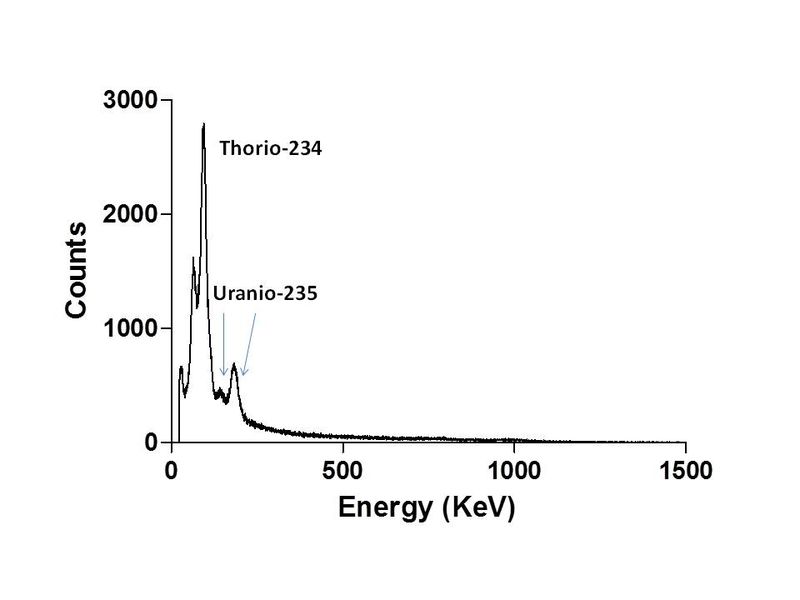
- In the spectrum of the radioactivity of the opal, why appears uranium-235 and other elements? if U-238 is the major isotope in natural uranium, where is it?. The U-235 is dangerous?
There is certain concern about uranium. Some people does not expect the presence of U-235, well known by their nuclear uses. Well, the gamma spectrum showed in my report is the typical radioactiviy spectrum of uranium in naturally occurring radioactive materials (NORM), as rocks or minerals. If we measure the spectrum of U in any mineral or rock, is always the same.
The main component in uranium, the U-238 (99.28% of natural uranium) is an element with a long half-life and, hence, a poor radioactive emitter. It is transmutated by alpha emission in Thorium-234. This transformation is accompanied by emission of gamma rays, but with a very low energy. Its energy is predated in the spectrum by the daughter nuclides and the X-ray emission. This is why, despite its abundance, the U-238 is invisible in this spectrum. The U-235 has a lower half-life and is a more intense emitter. The majority of radiation emitted natural uranium does not come from uranium, but from its daughter nuclides in the uranium series. These nuclides appears at constant concentration in NORM, because the uranium older than 700000 years is in "secular equilibrium". This is why the spectrum is the same in all minerals and rocks and this is why the uranium in minerals is more radioactive than purified uranium.
Do not be afraid by terms as uranium, X-ray, etc. This is a completely natural process and, in the quantities contained in opals, is completely safe.
- Is possible that the opals are a fake, produced by treatment of opals with uranium salts?
This is absolutely impossible. And, in the hypothetical case of a fake, the gamma spectrum would reveal the fraud, as the spectrum of uranium in laboratory chemicals or other processed materials is completely different.
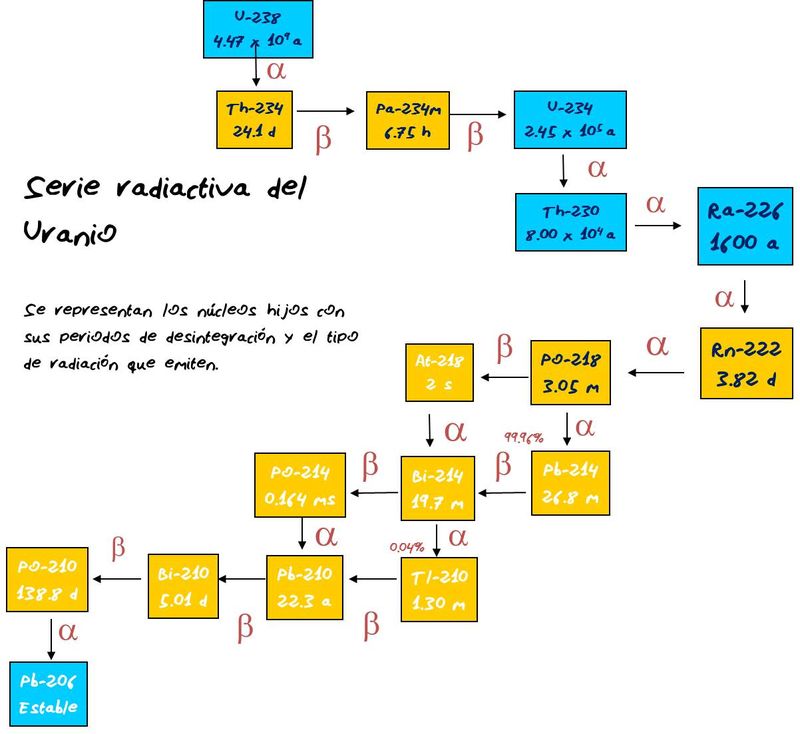
| Description: |
| The uranium decay series, which finish in the "radiogenic lead". The proportion between U and radiogenic lead is very useful in geologic dating |
|
| Viewed: |
23299 Time(s) |
|
| Description: |
| Spectrum of natural uranium after resetting (i.e., purification from other components) and waiting one year to gain equilibrium with its first daughter in the series. Note the peak of the 235 isotope. This isotope is purified for use in nuclear fuel and the residue is the "depleted uranium". |
|
| Viewed: |
23206 Time(s) |
|
|
|
| Back to top |
|
 |
Peter Megaw
Site Admin

Joined: 13 Jan 2007
Posts: 963
Location: Tucson, Arizona



|
 Posted: Mar 18, 2014 14:12 Post subject: Re: Daylight Fluorescent Hyalite Opal from Mexico Posted: Mar 18, 2014 14:12 Post subject: Re: Daylight Fluorescent Hyalite Opal from Mexico |
|
|
I have been playing around with another piece of this material and have taken a series of new pictures showing the different fluorescent responses to various lighting conditions. (Incandescent W light; daylight; a combination of Incandescent and SWUV and full SWUV).
I discovered that there appears to be a distinct difference in the daylight fluorescence between early morning (8:00) versus early afternoon (14:00); the later is stronger and yellower than the early shot. Both were taken in the same place under full shade with otherwise unrestricted view to the sun. No cloud cover whatsoever. The only real difference in the photographic conditions seems to have been the sun's position...and of course amount of atmospheric filtering.
All the photographs are of the same piece
| Description: |
Hyalite Opal
Central Mexico
170 x 100 mm
Hyalite opal photographed under 100 watt W incandescent light |
|
| Viewed: |
23067 Time(s) |

|
| Description: |
Hyalite Opal
Central Mexico
170 x 100 mm
Hyalite opal photographed under full shade at 8 am |
|
| Viewed: |
23059 Time(s) |

|
| Description: |
Hyalite opal
Central Mexico
170 x 100 mm
Hyalite opal photographed under full shade at 14:00 in exactly the same place as 8 am shot |
|
| Viewed: |
23057 Time(s) |

|
| Description: |
Hyalite Opal
Central Mexico
170 x 100 mm
Hyalite opal photographed with a combination of W Incandescent light and SWUV |
|
| Viewed: |
23062 Time(s) |

|
| Description: |
Hyalite Opal
Central Mexico
170 x 100 mm
Hyalite Opal photographed under SWUV |
|
| Viewed: |
23060 Time(s) |

|
_________________
Siempre Adelante! |
|
| Back to top |
|
 |
Vinoterapia
Joined: 03 Feb 2009
Posts: 179
Location: Houston, Tx



|
 Posted: Mar 19, 2014 08:59 Post subject: Re: Daylight Fluorescent Hyalite Opal from Mexico Posted: Mar 19, 2014 08:59 Post subject: Re: Daylight Fluorescent Hyalite Opal from Mexico |
|
|
| Amazing, this really is a kind of "Chameleon" mineral.
|
|
| Back to top |
|
 |
waterdog
Joined: 31 Dec 2012
Posts: 10


|
 Posted: Mar 24, 2014 22:19 Post subject: Re: Daylight Fluorescent Hyalite Opal from Mexico Posted: Mar 24, 2014 22:19 Post subject: Re: Daylight Fluorescent Hyalite Opal from Mexico |
|
|
Our specimen arrived today, thanks Jordi!
I checked the specimen with a Ludlum Type 3 radiation detector with a Model 44-9 pancake probe. The probe picks up alpha/beta/gamma. The response was 600 counts per minute at 0.5 cm from the detector face. The sample covered about 70% of the detector face. This count translates to 0.2 mrem/hour (2 μSv/hour), a worthless number for actual dose calculations but it gives a crude estimate of the radioactivity of the sample. The sample clearly radiates above background (less than 50 counts per minute in a dusty room). The specimen kicks out 18 mSv/yr while the average annual radiation dose (in US) is 6.2 mSv/year. So don't keep one in your pocket. :)
P.S. The meter hasn't been calibrated in 20 years.
|
|
| Back to top |
|
 |
Clifford Trebilcock

Joined: 14 Mar 2014
Posts: 12
Location: Phippsburg, Maine



|
 Posted: Mar 28, 2014 21:55 Post subject: Re: Daylight Fluorescent Hyalite Opal from Mexico Posted: Mar 28, 2014 21:55 Post subject: Re: Daylight Fluorescent Hyalite Opal from Mexico |
|
|
Hi Peter,
I have been following this topic with great interest. Lots of nice photos and detailed information. I have collected very similar Hyalite Opal specimens from the Consolidated Quarries here in Topsham, Maine. The Hyalite Opal coating is not quite as thick as the specimens you show from Mexico, but reacts exactly the same as the specimens from Mexico. I just assumed the Hyalite contained enough Autunite inclusions to fluoresce and give it the yellow/green color from the UV in the sunlight. I picked up several specimens without the use of UV light, and noticed the color was very hard to see in incandescent light but never paid much attention. Thin coatings of Hyalite Opal are pretty common in Maine pegmatites. Most is white or colorless and very hard to spot without a UV light. Some Hyalite Opal takes on a blue color with UV fluorescence decreasing as the blue color increases. I would guess trace amounts of copper are responsible for this material.
Cliff
|
|
| Back to top |
|
 |
jorgedavid
Joined: 10 Feb 2008
Posts: 17
Location: Mexico City



|
 Posted: May 06, 2015 19:10 Post subject: Re: Daylight Fluorescent Hyalite Opal from Mexico Posted: May 06, 2015 19:10 Post subject: Re: Daylight Fluorescent Hyalite Opal from Mexico |
|
|
| Peter Megaw wrote: | I picked up some interesting new material here at Tucson the other day
it is Hyalite opal from somewhere (Jalisco?) in central Mexico that shows what appears to be daylight fluorescence
Indoors under fluorescent or incandescent light it has almost no color at all (picture 1) but in daylight it has an attractive "'uranyl" yellow-green color (picture 2) similar to, but not as intense as the strong color it shows under Short-Wave UV (picture 3).
I have heard of similar material from Erongo? but not seen it.
The best pieces of this material is completely transparent with smooth botryoidal surfaces and small free-standing druplets.
Hopefully we will be able to get more complete location information someday |
Hi Peter, Las Vigas, also is locality for fluorescent hyalite . The daylight color is lilac and fluorescence is when short uv is used. Saludos.
|
|
| Back to top |
|
 |
|


