| View previous topic :: View next topic |
| Author |
Message |
Screenname
Joined: 20 Feb 2010
Posts: 99
Location: Washington



|
 Posted: Feb 23, 2010 13:24 Post subject: Home Grown Crystals Posted: Feb 23, 2010 13:24 Post subject: Home Grown Crystals |
|
|
I know all of you know about grow your own crystal sets and I have never actually done this before and have been really looking into it lately and have acquired some great recipes but all home grown crystals don't last as they dissolve in high moisture areas or when they are wet. Any way to keep this from happening or does anyone know of a recipe for one that wont dissolve? And how came it's so easy to grow crystals like that but things like quartz require heat and pressure but all salt requires is a seed crystal, or ice just requires freezing water?
_________________
Daniel |
|
| Back to top |
|
 |
Carles Millan
Site Admin

Joined: 05 May 2007
Posts: 1531
Location: Catalonia



|
 Posted: Feb 23, 2010 14:04 Post subject: Re: Home Grown Crystals Posted: Feb 23, 2010 14:04 Post subject: Re: Home Grown Crystals |
|
|
| Screenname wrote: | | And how came it's so easy to grow crystals like that but things like quartz require heat and pressure but all salt requires is a seed crystal, or ice just requires freezing water? |
There are a lot of chemical and physical differences between quartz and the substances you mention. Salt is an ionic compound, readily soluble in water. Ice, that is actually a mineral, has, as even kids know, a very low melting point temperature, about 273K. Quartz is neither water soluble nor melts at a low temperature (its melting point being 1883K or 1610ºC). It is not an ionic compound neither, since has a strong covalent bond between the silicon and the oxigen, not like the rather loose one that keeps the sodium chloride atoms together.
Many, if not most, quartz deposits are of hydrothermal origin. For instance, Hot Springs, Arkansas, USA. This means that the crystals were grown from an aqueous solution. But quartz being almost insoluble in water (in normal conditions), very high temperatures and pressures were needed for that happening. The critical temperature of water is 647K (or 374ºC) and its critical pressure 22MPa (or 218 atmospheres). In such conditions, that were common many millions of years ago and perhaps even today very deep in the Earth, quartz becomes easily soluble and crystallization from water in clefts takes place caused by oversaturation when either the temperature or the pressure (or both) decrease.
Hope I helped.
_________________
Al carrer Duran i Bas, si no hi vas no t'hi duran |
|
| Back to top |
|
 |
Pete Richards
Site Admin

Joined: 29 Dec 2008
Posts: 843
Location: Northeast Ohio



|
 Posted: Feb 24, 2010 21:45 Post subject: Re: Home Grown Crystals Posted: Feb 24, 2010 21:45 Post subject: Re: Home Grown Crystals |
|
|
We all are interested in minerals and crystals, but that does not mean that we all know about growing our own. That is a somewhat different topic. But certainly some of us have tried to grow our own, and maybe there's even an expert out there.... I'm not.
Carles Millan gave a very good technical answer for many aspects of your questions, but it may not be easy for you to understand, if you are not scientifically trained. To his comments I would add the following, attempting to avoid scientific complexity:
Hoping not to simplify too much, I would say that you are writing about crystals that can be grown from materials dissolved in water (salt, sugar, alum, copper sulfate, etc) at the temperatures and pressures of the earth's surface. The crystals grow because we change something (cooling a warm solution, evaporating some of the water) which leaves too much of the dissolved substance in the water, and the result is that one (or many) crystal(s) form(s) and grow until the imbalance is removed. If we grow crystals by evaporation, continuing evaporation maintains the imbalance, and the crystal can grow for a longer time and get larger.
This process is reversible. If you add more water to the jar in which you're growing the crystal, or warm it up, some (or all!) of the crystal will dissolve to re-establish the balance between material dissolved in the water and the same material as a crystal.
If you grow a crystal, and take it out of the solution, and keep it dry, it can remain unchanged for a long time. But if you get it wet, some of it will dissolve. Some crystals attract water from the air; they can "melt" in the water they attract. Some crystals have a certain amount of water in their structure; if that water evaporates away in a dry environment, the crystal structure can be disrupted and they turn to dust. Home-grown crystals of some chemicals do not have these problems, those of other chemicals do. To prevent these problems, the best non-technical approach is to isolate the crystal from the environment - keep it in a little sealed jar or a plastic bag. This may not stop the destructive processes, but it can slow them down greatly.
Some people would coat the crystal with oil or lacquer, but I don't recommend that because it changes the appearance of the crystal to something quite artificial.
Whatever you do, if your home grown crystals get dusty, don't rinse them off under the tap!
_________________
Collecting and studying crystals with interesting habits, twinning, and epitaxy |
|
| Back to top |
|
 |
Screenname
Joined: 20 Feb 2010
Posts: 99
Location: Washington



|
 Posted: Feb 25, 2010 10:48 Post subject: Re: Home Grown Crystals Posted: Feb 25, 2010 10:48 Post subject: Re: Home Grown Crystals |
|
|
I have yet to find a recipe for making one out of copper sulfate, does that one dissolve? It almost sounds like it would turn to metal.
_________________
Daniel |
|
| Back to top |
|
 |
Carles Millan
Site Admin

Joined: 05 May 2007
Posts: 1531
Location: Catalonia



|
 Posted: Feb 25, 2010 10:58 Post subject: Re: Home Grown Crystals Posted: Feb 25, 2010 10:58 Post subject: Re: Home Grown Crystals |
|
|
| Screenname wrote: | | I have yet to find a recipe for making one out of copper sulfate, does that one dissolve? It almost sounds like it would turn to metal. |
There are in the market a number of inexpensive kits to easily grow very nice crystals with vivid colors. I remember having bought one for my son in a NYC gift shop back in the mid nineties. And it was fun! Many natural history museums also sell them.
Just google the words "crystal growing kit".
Good luck!
|
|
| Back to top |
|
 |
Screenname
Joined: 20 Feb 2010
Posts: 99
Location: Washington



|
 Posted: Feb 25, 2010 12:19 Post subject: Re: Home Grown Crystals Posted: Feb 25, 2010 12:19 Post subject: Re: Home Grown Crystals |
|
|
Hey, I was having funning googling this topic and discovered something different. Alcohol crystals, making crystals with alcohol instead of water and these are less soluble. Any ideas on this?
_________________
Daniel |
|
| Back to top |
|
 |
Paul S

Joined: 20 Mar 2010
Posts: 79



|
 Posted: Mar 27, 2010 18:24 Post subject: Re: Home Grown Crystals Posted: Mar 27, 2010 18:24 Post subject: Re: Home Grown Crystals |
|
|
Growing crystals yourself is a fantastic hobby. I do it alot and it gets me excited every time I pull a nice crystal out of a solution or liquid. I could keep on talking about making synthetic crystal all night long, but I don't think it's the right place for it.
There are a few interesting things you might try though:
- mothballs (containing naphtalene or 1,6-dichlorbenze) will give nice crystals. put some balls in a glass jar and close it properly with a lid. You want the gasses to stay in. Now heat the jar in a bowl of hot water. The mothballs will melt. Let it cool down and watch the crystals grow on the inside of the jar. You can open the jar if you like, the crystals are fragile and small though, but they are genuine gas-phase deposition grown crystals!
- sodium acetate can be turned into a supercooled liquid. If you then put a litle piece of sodium acetate crystal in the liquid it will crystalize instantaneously. the same stuff is used in those handwarmers you might have come across some time. This can also be done with sodium thiosulfate.
About 'alcohol crystals': they are just like normal crystals, but you can use other materials than with water soluble crystals. Not every chemical can be dissolved in water, but sometimes they can be dissolved in alcohol. You grow them however in the same way as you would grow water soluble crystals: by evaporating a saturated solution or by cooling down a supersaturated solution.
About the lifespan of crystals, it really depens on the chemicals they are made from. I have some chrome alum crystals that started dehydrated after a few weeks and turned into a light purple powder. Coppersulfate can do the same, but takes some more time. I have however a red prussiate crystals with me that has been lying on my shelve without any protection for more than a year now, without any change in its appearance.
Cleaning crystals can best be done with a very soft brush or with compressed air from a can. Never with water, unless they are ofcourse insoluble. Still, I would only use water on metal crystals, and not even tap-water but demineralised water to prevent any staining or reactions.
There is also the option of coating crystals to prevent them from deteriorating. the best thing to use is a two-component resin. They will not dissolve the crystals as some varnishes or glues will. I use stuff that is used for a process called 'cold glazing'. It is very thin so it will not fill up corners too much. It is however inevitable that the crystals will look a little 'plastic' after the treatment. It works well if you want to use the crystal for making jewellery, because it protects them against sweat and scratching.
You can always contact me at: paul(at)paulslab(dot)com if you want to know more about growing crystals yourself. Always inform yourself about experiments and chemicals, safety first!
|
|
| Back to top |
|
 |
kakov

Joined: 24 Jul 2013
Posts: 39
Location: Madrid



|
 Posted: Jan 01, 2014 19:25 Post subject: Re: Home Grown Crystals - sodium acetate Posted: Jan 01, 2014 19:25 Post subject: Re: Home Grown Crystals - sodium acetate |
|
|
I tried today the one with growing sodium acetate.
| Description: |
sodium acetate (home grown)
locality: n/a
surface 14 x 22 cm, 0,5 cm thick layer
It got fully crystallized in around 15 seconds, during which you can see the crystals growing, it is really impressive! |
|
| Viewed: |
54419 Time(s) |

|
| Description: |
sodium acetate (home grown)
locality: n/a
section from full surface of 14 x 22 cm
Everything was done in a normal kitchen.
I used distilled water but it might also work with water from the tap.
The sodium acetate was brought in Manuel Riesgo in Madrid. |
|
| Viewed: |
54482 Time(s) |

|
|
|
| Back to top |
|
 |
Mark Beregszaszi
Joined: 26 Dec 2013
Posts: 33
Location: Budapest


|
 Posted: Jan 12, 2014 21:29 Post subject: Re: Home Grown Crystals Posted: Jan 12, 2014 21:29 Post subject: Re: Home Grown Crystals |
|
|
I have grown potassium-alum, Ca-acetate, Na-acetate, Copper-sulphate, Iron-sulphate, Copper-acetate, Native silver crystals mostly at home.
Will be experimenting with chrome alum too.
The easiest are the potassium alum and copper sulphate crystals and supposedly chrome alum. You can dye potassium alum with chrome alum.
| Description: |
Ca-acetate grown on scarn
Home-grown
About 5cm wide.
Put a piece of carbonate bearing rock in 20% acetic acid. The acid should cover the stone.
Let it stay still until the liquid evaporates and this structure will grow upwards on the stone. Very interesting, even kids can do it :) |
|
| Viewed: |
54171 Time(s) |

|
| Description: |
Ca-acetate
Home-grown
Crytal needles up to 3cm in length.
Grown from calcite + acetic acid. |
|
| Viewed: |
54216 Time(s) |

|
| Description: |
Ca-acetate
Home-grown
Up to 5cm in height
Calcite + acetic acid |
|
| Viewed: |
54172 Time(s) |

|
| Description: |
Cu-acetate (Dark, almost black), Cu-sulphate(blue), Na-acetate(no color)
Home-grown
Cu-acetate crystals up to 1cm.
Just a home experiment.
Dissolved soda in acetic acid -> Na-acetate.
Added Cu-sulphate.
Some Cu-acetate crystals formed.
Still cheaper to buy pure Cu-acetate in the shop. |
|
| Viewed: |
54125 Time(s) |

|
| Description: |
Native silver with a little copper coating on a few crystals
Home-grown
Crystals up to 1cm
Grown from Ag-nitrate solution on a piece of copper coin. |
|
| Viewed: |
54182 Time(s) |

|
| Description: |
Potassium alum
Home-grown
In an 5l jar..
Brownish because I tried to color with KMnO4. Did not work well. |
|
| Viewed: |
54203 Time(s) |

|
| Description: |
Potassium alum
Home-grown
Hand for comparison.
My favorite pot. alum crystal.
At the local mineral club many thought it was a killer chinese fluorite :) |
|
| Viewed: |
54338 Time(s) |

|
| Description: |
Materials for growing
Home
..
Pot. alum, CuSO4, FeSO4, etc.. |
|
| Viewed: |
54152 Time(s) |

|
| Description: |
Na-acetate
Home-grown
Length up to 3cm
Na acetate crystals from the batch I made for my Cu-accetate experiment. |
|
| Viewed: |
54202 Time(s) |

|
| Description: |
Potassium alum, another photo of my big crystal.
Home-grown
Same size as the previous pic. |
|
| Viewed: |
54167 Time(s) |

|
|
|
| Back to top |
|
 |
kakov

Joined: 24 Jul 2013
Posts: 39
Location: Madrid



|
 Posted: Jan 13, 2014 16:22 Post subject: Re: Home Grown Crystals Posted: Jan 13, 2014 16:22 Post subject: Re: Home Grown Crystals |
|
|
Thanks a lot Mark for sharing these exciting experiments with us.
If you do not mind, could you please give some hints and tips regarding how you got such excellent results with the potassium alum. Many thanks.
(PS, Just in case, I hope nobody have any nasty second thoughts around this string. The reason for growing our own crystals is not to fake natures wonders rather to learn more about them. A direct experience with these simple crystallization processes might enrich our experience in watching the real stuff and imagining how it could have grown.)
|
|
| Back to top |
|
 |
Mark Beregszaszi
Joined: 26 Dec 2013
Posts: 33
Location: Budapest


|
 Posted: Jan 13, 2014 17:49 Post subject: Re: Home Grown Crystals Posted: Jan 13, 2014 17:49 Post subject: Re: Home Grown Crystals |
|
|
Of course :)
I grow crystals, because they can turn out to be real beauties and the whole crystallization proccess is very interesting.
First you should get a few kgs of potassium alum.
Best if you buy it at a painting and building material shop. Here it is around 5euro/kg. (do not know the proper word for it in English...)
You should be able to work with 2-3kg I think, but i havent been growing crystals for some time, do not remember exactly. The more material you have the better, it will not go to waste, you can always dissolve them in water and recrystallize again.
If you have your potassium alum, pick a cooking pot that is covered with enamel.
Pick a glass jar that you can close with a cap.
Put at least as much water in the cooking pot as much water fills the jar and heat the water until it starts to boil.
Then add you potass alum to it until no more dissolves.
Then pour the hot solution into the glass jar, close it tight.
Wrap the jar with clothes and pillows, the slower it cools down the better.
After about 2 days when it is cold you can open the jar, pour out the liquid and see your crystals at the bottom.
Carefully break up those crystals, pick one big piece that is one big crystal fragment and tie a thin string or wire on it. This will be your seed crystal.
Repeat the dissolving proccess with your salt, but keep the one crystal piece you have on a string.
When your solution does not dissolve more salt, pour it on the jar and hang your seed crystal into it from the cap of the jar. (make a hole in the center of the cap, pull the string through the hole and fix it on the top.)
So your seed crystal sould be hanging into the liquid and it should not dissolve if you did it right.
let is SLOOOOOWWWLLLYYY cool down and if you are lucky you will have a big single crystal grown of your seed crytals, the rest of the crystals grown on the bottom of the jar.
If you want a big crystal like mine, you have to use a bigger jar with more solution.
I hope this is understandable, when I have enough time I can make a little tutorial for it in Paint, but that will be in february the soonest.
|
|
| Back to top |
|
 |
Mark Beregszaszi
Joined: 26 Dec 2013
Posts: 33
Location: Budapest


|
 Posted: Jan 14, 2014 07:14 Post subject: Re: Home Grown Crystals Posted: Jan 14, 2014 07:14 Post subject: Re: Home Grown Crystals |
|
|
How to grow Ca-acetate needles.
1: Crush calcite with a hammer
2: Put the calcite pieces in a plastic cup, fill the cup with acetic acid (20%)
3: Let it stay for 2 days, then pour the liquid into another cup.
4: Wait until the liquid evaporates, you will have a white disk left in the bottom of the cup
5: Take out the white disk and cut it in half
6: Blow some air at the cut surface to separate needles.
If you are lucky you will not not have to cut the disk, but you will be able te remove the top layer and uncover better crystals.
Have fun :)
| Description: |
| How to make Ca-acetate needles |
|
| Viewed: |
53968 Time(s) |

|
|
|
| Back to top |
|
 |
Turbo
Joined: 30 Sep 2009
Posts: 260
Location: Delaware



|
 Posted: Jan 15, 2014 14:06 Post subject: Re: Home Grown Crystals Posted: Jan 15, 2014 14:06 Post subject: Re: Home Grown Crystals |
|
|
I grow copper sulfate pentahydrate as a hobby. The natural occurrence of this mineral is called chalcanthite. I grow these through a combination of cooling a supersaturated solution and evaporation. You can see the variety of crystals here because I can control how long they grow, use seed crystals for preferential growth, or graft crystals from one onto another.
These crystals will stay looking pretty as long as they are kept out of excessively humid or dry conditions because not only do they dissolve easily, they are also hygroscopic and will therefore lose or gain water depending on the environment. I keep mine in closed containers. I've seen no breakdown over the course of a year.
| Description: |
Copper sulfate pentahydrate
Lab grown
largest is 12cm across |
|
| Viewed: |
53836 Time(s) |

|
| Description: |
Copper sulfate pentahydrate
lab grown
miniatures
Copper sulfate crystals stored in plastic container. |
|
| Viewed: |
53888 Time(s) |

|
|
|
| Back to top |
|
 |
Mark Beregszaszi
Joined: 26 Dec 2013
Posts: 33
Location: Budapest


|
 Posted: Jan 15, 2014 15:02 Post subject: Re: Home Grown Crystals Posted: Jan 15, 2014 15:02 Post subject: Re: Home Grown Crystals |
|
|
| Nice crystals and thanks for the idea, never thought about grafting before :)
|
|
| Back to top |
|
 |
Cesar M. Salvan
Site Admin
Joined: 09 Jun 2008
Posts: 127
Location: Alcalá de Henares



|
 Posted: Jan 15, 2014 17:11 Post subject: Re: Home Grown Crystals Posted: Jan 15, 2014 17:11 Post subject: Re: Home Grown Crystals |
|
|
I think that this thread in the forum is a nice way to celebrate the Year of Crystallography.
Here I go with my contributions. I, as a microscopist, tend to put my eyes in small crystals...so, fans of big crystals, forgive me.
| Description: |
Lead
Home grown
Weird habit of lead crystals developed by electrochemical reduction of a Pb(II) solution. |
|
| Viewed: |
53716 Time(s) |

|
| Description: |
Gypsum
Home grown
Gypsum crystal developed by gel crystallization. The technique of gel crystallization is very interesting and useful in the modelization of the habit preference during formation of certain minerals. |
|
| Viewed: |
53836 Time(s) |

|
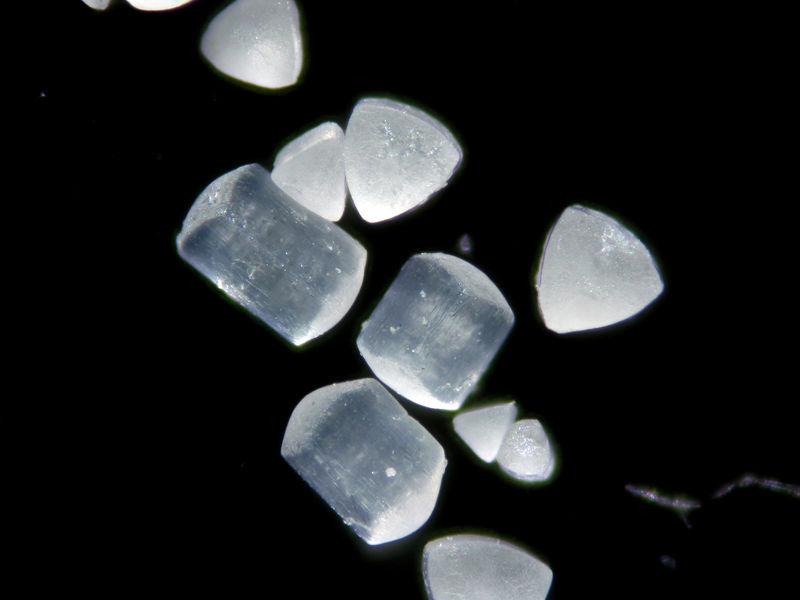
| Description: |
Anhydrite
Home grown
Home grown anhydrite (phase confirmed by DRX). Note the bluish color. |
|
| Viewed: |
53780 Time(s) |
|
| Description: |
Alanine
Home grown
Synthetic alanine (the second simplest amino acid), obtained during Prebiotic Chemistry experiments, crystallized by slow evaporation. Pretty out of the neighborhood of the amateurs, but still "home grown", isn't it? |
|
| Viewed: |
53795 Time(s) |

|
| Description: |
Ice
Home grown
Is uncommon to see hexagonal prisms of Ice. The crystals of the picture were obtained by sublimation of ice at - 20ºC and low pressure. The picture was taken under great stress: my microscope was not equipped with low temperature stuff... |
|
| Viewed: |
53727 Time(s) |

|
|
|
| Back to top |
|
 |
|





